Heat Treatment of Steels
Conventional heat treatments for Hypo- and Hyper-eutectoid steels. The goal is to modify microstructure (Grain size, phase distribution) to alter mechanical properties.
Reminder from Non-Equilibrium Phase Diagram
- : Eutectoid temperature (~727°C). Boundary between Austenite and Pearlite.
- : Upper critical temperature for Hypo-eutectoid steels (Austenite limit).
- : Upper critical temperature for Hyper-eutectoid steels (Cementite solubility limit).
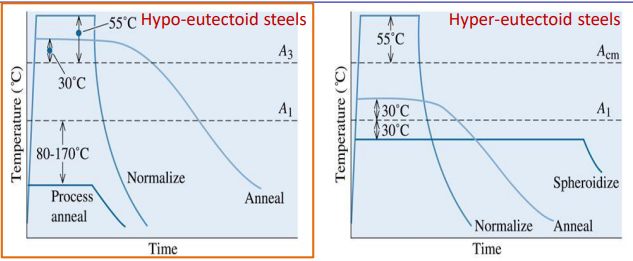
Hypo-Eutectoid Steels ()
Normalizing
- Process: Heat to (Austenitization) Air Cool.
- Result: Moderate grain refinement.
- Properties: Harder and stronger than annealed; good mechanical properties at low cost.
- Microstructure: Fine Pearlite + Ferrite.
Full Annealing
- Process: Heat to Furnace Cool (Very slow).
- Result: Maximum chemical homogenization and softening.
- Properties: Maximum ductility, lowest hardness.
- Microstructure: Coarse Pearlite + Ferrite.
Process Annealing
- Process: Heat to below .
- Result: Recrystallization of ferrite without phase transformation.
- Use Case: Restoring ductility after cold working (strain hardening recovery).
Hyper-Eutectoid Steels ()
Normalizing
- Process: Heat above Air Cool.
- Goal: Break up the brittle Cementite network at grain boundaries.
Spheroidizing
- Process: Pendulum heating (oscillating) around .
- Result: Spheroidization of Pearlite and Cementite.
- Properties: The softest, toughest possible condition for high-carbon steel.
- Use Case: Essential for machinability of high-carbon steels.
Overview of Heat Treatments
- Austenitization: Holding at to dissolve C into -Fe (max solubility/homogeneity).
- Annealing: Diffusion-driven. Max softening. Equilibrium microstructure.
- Normalizing: Air-cooled. Slightly non-equilibrium. Moderate refinement.

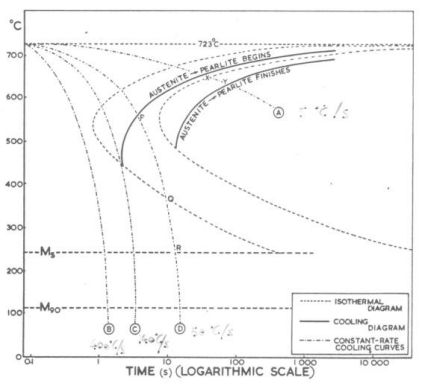
Isothermal H.T. On a TTT/CCT Diagram
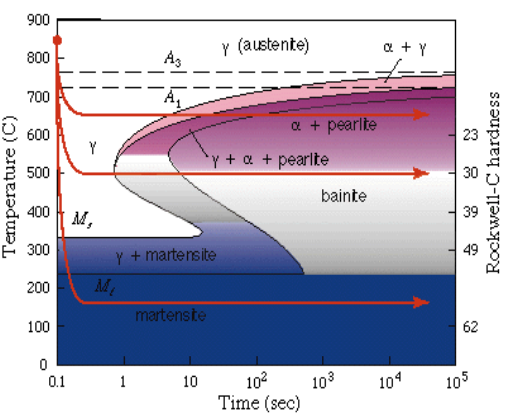
Bainite
A microstructure of Ferrite and Cementite that forms between the temperature ranges of Pearlite and Martensite (roughly 250°C – 550°C). While martensite is BCT, bainite is BCC.
Formation: Created via Austempering (isothermal holding). You cool the steel fast enough to miss the Pearlite nose, but hold it above the Martensite start () line until it fully transforms.
The Two Types:
- Upper Bainite (400°C – 550°C): Forms at higher temps. Looks “feathery.” The carbides precipitate between the ferrite plates. It’s tough, but lower bainite is usually better.
- Lower Bainite (250°C – 400°C): Forms at lower temps. Looks like needles (acicular). The carbides precipitate inside the ferrite needles. High strength and high toughness.
Why it matters: It offers a unique balance. You get hardness similar to tempered martensite but with better ductility and toughness. Plus, since you don’t do a violent quench to room temperature, you avoid the internal stresses and cracking risks associated with Martensite.
Note
TTT (Time-temperature-Transformation) diagrams show transformations in isotermal environments. In the real world, this is impossible, since materials cannot change temperature instantly. CCT (Continuous Cooling Transformation) diagrams show the real world since things cool down gradually. Typically, reactions are slower than in an ideal world, so in a CCT diagram reactions are shifter bottom-right with respect to a TTT diagram.(reactions are slower and happen at lower temperatures).
Quenching
Quench Severity Coefficient aka. H-Value
Definition
Describes how aggressively a medium (the liquid the metal is quenched in) cools down the material, with 1 being the baseline and the value for still water.
$$$Q=HA\Delta T$$
| Quench Medium | Agitation? | H Coefficient | Cooling Rate (°C/s)* |
|---|---|---|---|
| Oil | No | 0.25 | 18 |
| Oil | Yes | 1.0 | 45 |
| Water () | No | 1.0 | 45 |
| Water () | Yes | 4.0 | 190 |
| Brine | No | 2.0 | 90 |
| Brine | Yes | 5.0 | 230 |
| *Cooling rate measured at the centre of a 1-inch bar. |
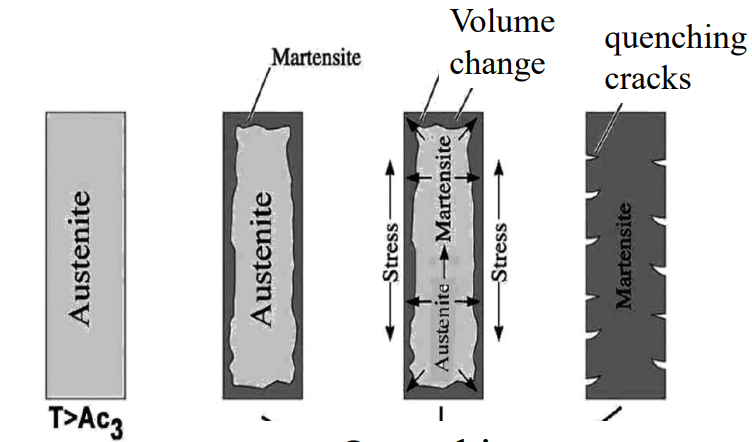
In a thick part, the surface cools much faster than the core. The surface transforms to Martensite (and expands) first, creating a hard shell. When the core later tries to expand, it is trapped, leading to high residual stresses and quench cracking. Quenching cracks are very common in larger pieces.
Marquenching: the solution to quenching cracks
An interrupted quenching method designed to minimize distortion.
- Quench: Rapidly cool to a temperature just above the Martensite Start () point.
- Hold: Keep it there until the temperature equalizes (Surface Temp Core Temp).
- Finish: Air cool through the Martensite transformation range.
Benefit: Since the temperature is uniform, the transformation happens simultaneously across the entire cross-section. This drastically reduces residual stress and cracking.
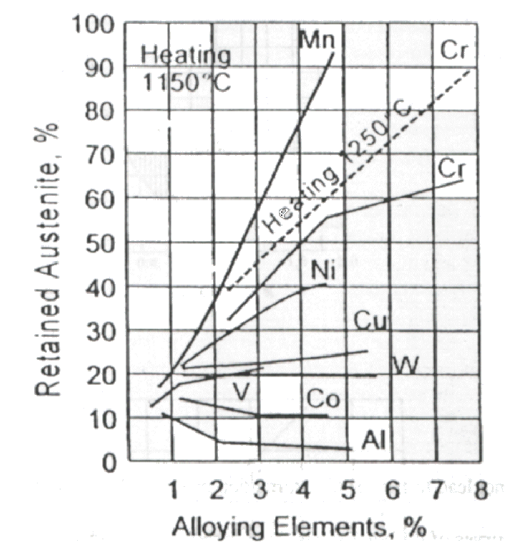
Residual Austenite ()
Definition
Austenite that fails to transform into Martensite during quenching because the quench didn’t get cold enough (specifically, the temperature didn’t drop below , the Martensite Finish temp).
Causes:
- High Carbon/Alloy Content: These elements push the temperature way down (often below 0°C).
- Stepped Quenching: Isothermal holding stabilizes the Austenite.
Consequences:
- Hardness & Fatigue Strength: Soft spots in the hard martensite matrix.
- Dimensional Instability: The residual will eventually transform later in service. This transformation causes Volumetric Expansion & Distortion.
- Internal Stresses: The delayed expansion creates new internal stress.
Solutions:
- Cryogenic Treatment: Continue quenching into sub-zero temperatures (Liquid Nitrogen at -150°C or Dry Ice at -80°C) to force the cross of the line.
- Multiple Tempering: Execute two or more Tempering cycles to decompose the retained austenite into stable phases.
Volumetric Expansion & Distortion
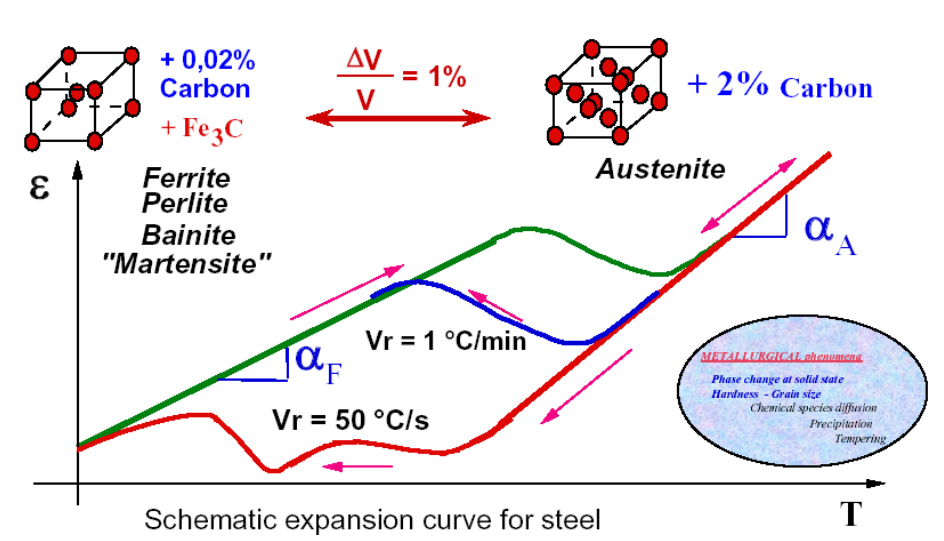
-
Thermal Contraction: As temperature drops, the metal wants to shrink.
-
Phase Transformation: As Austenite turns into Martensite (or Ferrite), the crystal lattice wants to expand.
-
Austenite (FCC): Ideally packed atoms. High density.
-
Martensite/Ferrite (BCC/BCT): Loosely packed atoms. Low density.
-
The Result: When the phase flips, the volume suddenly increases by roughly 1%.
This phenomenon can cause huge internal stresses, which can lead to quenching cracks or overwhelming deformations of the object.
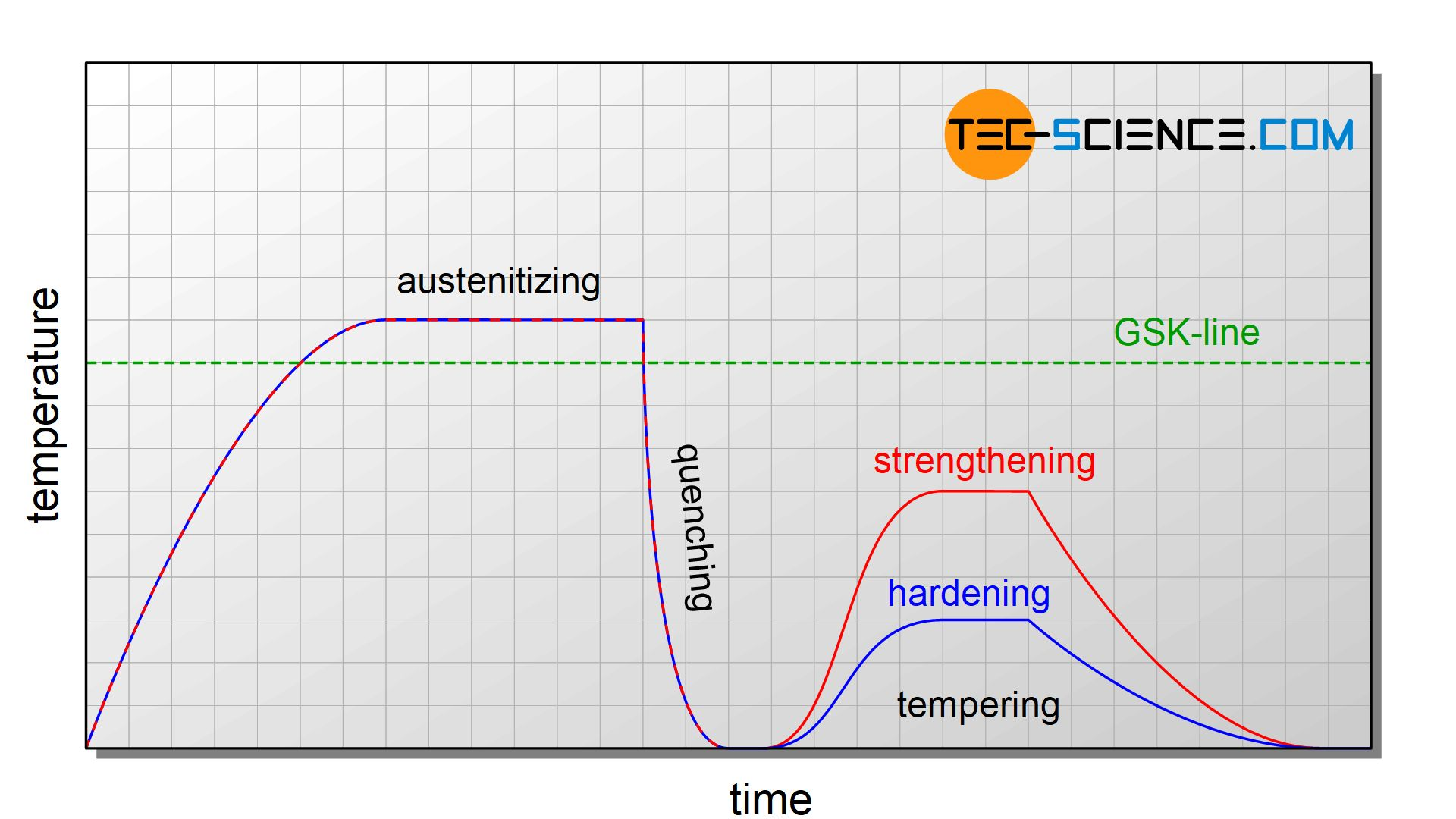
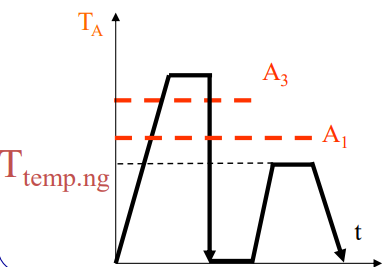
Tempering
Definition
Tempering is a heat treatment process, mainly for metals like steel, that involves reheating a hardened material to a specific temperature below its critical point and then cooling it slowly to reduce brittleness, increase toughness, and relieve internal stress, creating a better balance of strength and ductility for practical use.
Graph
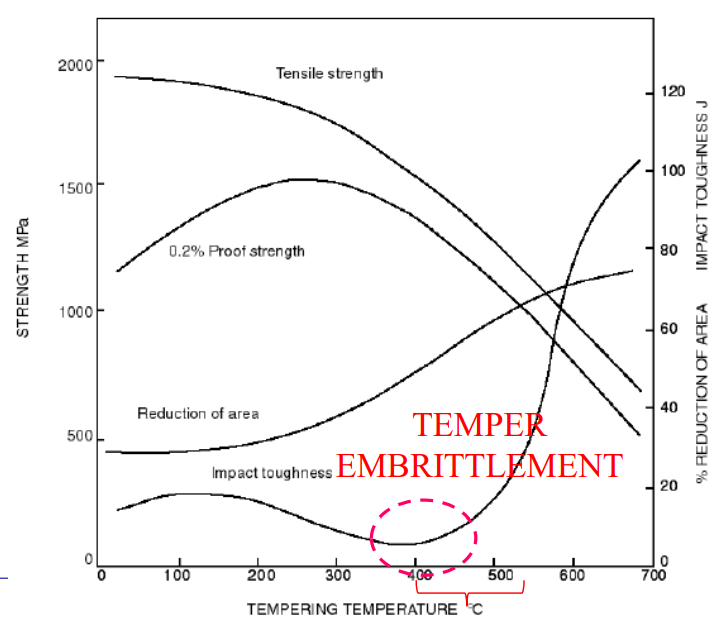
Temper Embrittlement (Krupp's Illness)
Normally, tempering increases toughness (). However, if you hold the steel in the 400°C – 550°C range (or cool slowly through it), the toughness drops dramatically.
You ruin the steel if you do any of these:
- Temper it specifically inside the 400-550°C range.
- Temper it above this range (>600°C) but then cool it slowly through the danger zone.
- Hold it in this range for too long during service.
The Result:
- Fracture Mode: Intergranular (the metal cracks along the grain boundaries because impurities segregate there).
- Reversibility: It is reversible. You can fix it by reheating above 600°C and quenching.
Who is Vulnerable?
- Safe: Plain Carbon Steels (with ) are immune.
- At Risk: Low alloy steels with high or .
- High Risk: Steels with coarse grains (more segregation).
How to fix it
- The Alloying Fix: Add (>0.2%) or (0.4%). This is why Chrome-Moly steel is great: the Molybdenum blocks the impurities from moving to the boundaries.
- The Process Fix: If you temper at high temps (>600°C), quench the part afterwards. Do not furnace cool it. You must “jump over” the 550-400°C gap quickly.
Why it Happens
- Impurities precipitate on grain boundaries at that temperature range
- combine with impurities, separating from the main alloy.
- are more stable in their carbide forms and therefore do not react with impurities.
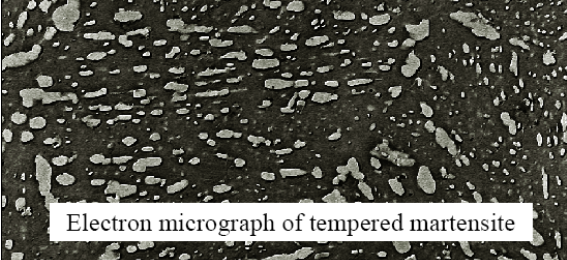
Tempered Martensite
Martensite is extremely hard and brittle. After tempering, the cementite phase turns into spheroidal shapes. The tempering is done at 250-650 °C. This reduces internal stresses and increases toughness.
Tempered Carbon Steels

| Element | Primary Roles | Advantages | Disadvantages | Notes |
|---|---|---|---|---|
| Hardener. | Yield Strength, Hardness (HRC), Hardenability | Toughness (CVN) | Combines with to form carbides. | |
| Hardenability Agent & Austenitizer. | Massive Hardenability (H), Strong Austenitizer (after ) | Ductility, Weldability (as concentration rises) | Role depends on %. | |
| Deoxidizer & Grain Refiner. | Powerful deoxidizer in the melt, refines grain size | Detrimental if > 0.1% | Used in “killed” steels. | |
| Deoxidizer & Elasticity Booster. | Elastic Limit, Deoxidizer (less than ) | Detrimental to surface finish if excessive | Less effective than for HRC. | |
| Corrosion Shield. | Corrosion Resistance (Rcorr) (if 0.2% - 0.5%) | Surface finish in hot-worked steels if excessive | Keep < 0.5%. | |
| Ferrite Strengthener & Toughener. | Tensile Strength (UTS) while maintaining high Toughness and Ductility. Strong Austenitizer | None | Stays in solid solution; best all-rounder. | |
| Best for high temperature. | Creep Resistance at high T, Hardenability (H) | None | Overcomes temper-embrittlement. | |
| Increases hardenability. | Hardenability (H) | None | - | |
| Grain Refiners & Hardeners. | Hardness (HRC) without depressing toughness, Wear Resistance, High T Resistance | None | Strong carbide formers and grain refiners. |