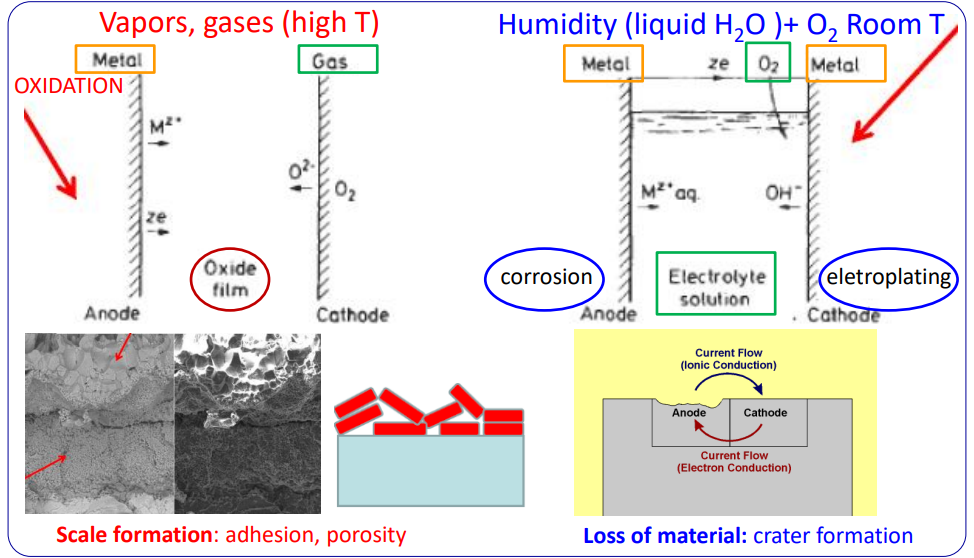
Dry vs. Wet Corrosion
Dry Corrosion (High Temperature Oxidation)
- Environment: Vapours and gases at High Temperatures.
- Mechanism: Direct chemical reaction between the metal and the gas (usually ).
- Process: .
- Outcome: Formation of an oxide film (scale).
- Adhesion and porosity of the scale determine if it protects the metal or allows further attack.
Wet Corrosion (Electrochemical)
- Environment: Humidity (liquid ) + Oxygen at Room Temperature.
- Mechanism: Electrochemical process involving an electrolyte.
- Components Required:
- Anode: Gives up electrons (Corrodes).
- Cathode: Accepts electrons (Protected).
- Electrolyte: Conducts ions ().
- Electrical Connection: Conducts electrons ().
- Outcome: Loss of material (crater formation) at the anode; often electroplating or byproduct formation at the cathode.
The Electrochemical Reactions
Stopping Wet Corrosion
For wet corrosion to occur, two simultaneous reactions must happen. If you stop one, you stop the corrosion.
The Anodic Reaction (Oxidation)
This is where the metal is lost. The metal dissolves into the electrolyte as ions. Examples:
The Cathodic Reaction (Reduction)
This occurs at the surface that does not corrode. The specific reaction depends on the environment’s pH and oxygen content.
| Environment | Condition | Reaction |
|---|---|---|
| Acidic | High ions | |
| Acidic | Dissolved | |
| Neutral/Basic | Dissolved |
Note: Metal ions can also be reduced (Electroplating): .
Classification of Corrosion Phenomena

1. Uniform Corrosion
- Appearance: Even removal of metal across the surface.
- Mechanism: Micro-corrosion cells shift randomly over time.
- Risk: Greatest destruction by mass, but least dangerous technically because it is predictable and easy to measure.
2. Galvanic (Bimetallic) Corrosion
- Mechanism: Two dissimilar metals in electrical contact in an electrolyte.
- The Law: The less noble (anodic) metal corrodes; the more noble (cathodic) metal is protected.
- Driving Force: The difference in electrical potential (Voltage) between the metals.
The “Area Ratio” Effect
The rate of corrosion is determined by the current density ().
- Big Cathode + Small Anode = DISASTER.
- The massive cathode demands a huge flow of electrons. The tiny anode must corrode furiously to supply them.
- Example: Steel rivets in a Copper plate Rivets disintegrate rapidly.
- Small Cathode + Large Anode = Safe.
- The corrosion is spread out over a large area and is sluggish.
3. Localized Corrosion
These are dangerous because the overall mass loss is low, but the structural damage is high.
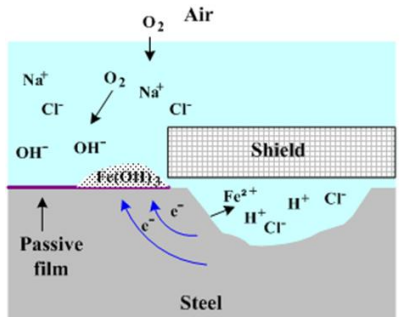
Crevice Corrosion
- Location: Narrow gaps (crevices) under bolts, gaskets, or deposits.
- Mechanism:
- Oxygen is depleted inside the crevice.
- The crevice becomes strictly Anodic (dissolution of metal).
- The area outside remains Cathodic (plenty of ).
- Positive metal ions accumulate in the crevice, attracting negative ions () and lowering pH (creating acid), which accelerates the attack.
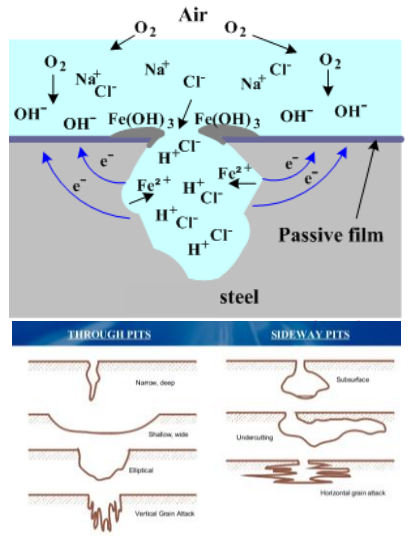
Pitting Corrosion
- Appearance: Small, deep holes.
- Mechanism: Similar to crevice corrosion but autocatalytic. Often initiated by Chloride ions () breaking the passive film.
- Danger: Occurs in materials that are otherwise passive (like Stainless Steel). It is insidious and hard to detect.
Synergistic Phenomena
Stress Corrosion Cracking (SCC)
- Formula: Tensile Stress + Specific Corrosive Environment + Susceptible Material.
- Mechanism: A crack initiates (frequently at a pit). The crack tip is active (anodic), while the walls are passive. The stress keeps opening the tip, exposing fresh metal to corrosion.
- Result: Sudden brittle failure in ductile materials.
Hydrogen Embrittlement
- Mechanism: Atomic Hydrogen () diffuses into the metal lattice (interstitial).
- Sources of H: Corrosion reactions, welding, electroplating.
- Effect: Low ductility fracture. Can be confused with SCC, but strictly due to Hydrogen presence preventing plastic deformation (dislocation movement).
Others
- Corrosion Fatigue: Fatigue limit is lowered (or eliminated) by a corrosive environment.
- Selective Leaching: One element is removed (e.g., Zinc removed from Brass).
- Fretting Corrosion: Corrosion assisted by small-scale vibration/sliding.
- Erosion Corrosion: Flowing fluid strips protective films.

The Galvanic Series (Seawater)
| Rank | Metals | Role |
|---|---|---|
| Noble (Cathodic) | Platinum, Gold, Graphite, Titanium | Protected (Will eat other metals) |
| Passive | Stainless Steel (Passive), Nickel (Passive) | |
| Active | Steel, Iron, Aluminum Alloys | |
| Sacrificial (Anodic) | Zinc, Magnesium | Corrodes (Used to protect others) |
Design Rule
Never couple a metal from the top of this list with a small piece of metal from the bottom.
Prevention and Protection
Design Rules
- Weld rather than rivet (avoids crevices).
- Drainage: Design containers to drain completely (avoid standing liquid).
- Stress: Avoid residual tensile stresses in exposed components (prevents SCC).
- Galvanic: Insulate dissimilar metals or keep them far apart in the series.
- Heterogeneity: Avoid varying conditions (heat, stress, aeration) across the surface.
Coatings & Active Protection
- Sacrificial Anodes: Connect the steel to Zinc or Magnesium. The Zn/Mg corrodes (anode), forcing the steel to be the cathode (immune).
- Passivation: Relying on stable oxide films (e.g., on Stainless Steel).
- An oxide layer forms on the surface, leading to decreased conductivity and therefore slower corrosion.
- Warning: If the film is damaged and cannot reform (no Oxygen), pitting occurs.
- Coatings: Paints, polymers, or metal plating (Cladding, Hot-dipping).