Humé-Rothery Rules for Alloy Formation
- Dimensional Factor
- Atoms tend to pack Atomic radii of solvent atoms must stay within 15% of solutes’
- Identical crystal structure of solute and solvent (e.g. BCC, FCC)
- Similar electronegativity of solute and solvent
- Valence Factor
- A solute will enter into a solution if its valence is greater that of the solvent’s
Condon-Morse Model
Important
The Condon-Morse model describes the relationship between intermolecular distance and their forces and potential energy.
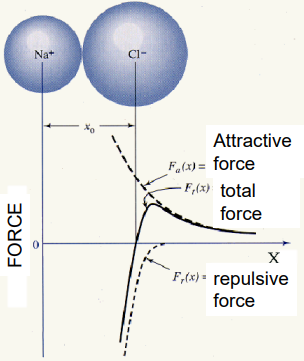
When we have an equilibrium, where there is no net force between the particles and we therefore have stability. As we increase distance above this point, intermolecular forces begin to pull the particles together (such as Van der Waals, ionic bonds, covalent bonds, external stress etc.), but if the particles get too close, we get overlapping electron clouds which cause repulsive forces.
Remember
Where:
- is the atomic distance
- is the attractive potential energy
- is the repulsive potential energy
Bonding energy is the energy required to separate two atoms: